Ionic Compounds & Chemical Nomenclature
chapter 7: ionic compounds and metals
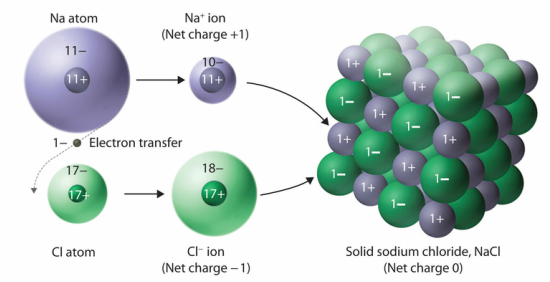
An ion is formed when an atom gains or loses one or more valence electrons in order to achieve a stable electron configuration. Oppositely charged ions attract each other, forming electrically neutral ionic compounds. When writing the name or formula of an ionic compound, the cation appears first and the anion follows. An example of this is salt: sodium is the cation while chloride is the anion, so it is written as sodium chloride or NaCl formulaically. Metals form crystal lattices and can be modeled as cations surrounded by a "sea" of freely moving valence electrons. This property makes metal alloys good conductors of electricity.
|
|
|
|