Atomic structure and nuclear chemistry
chapter 4: the structure of the atom
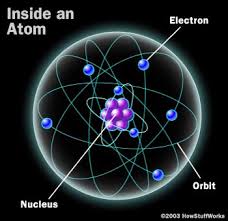
Although the ancient Greeks had many creative ideas about matter, our current understanding is based upon John Dalton's atomic theory, published in 1803. Essentially, matter is made up of atoms, which are indivisible and indestructible. All atoms of a given elements are the same in chemical properties and mass while compounds can be formed by using two or more different elements. Compounds can vary in properties as opposed to the elements that make them up. Despite varying historical depictions of the structure of the atom, we know know that the atom consists of a nucleus – which contains protons and neutrons – with electrons moving around in electron shells. The amount of protons and atomic mass determine the type of element. Unstable atoms will emit radiation.
|
|
|
|